 An atom consists of a dense nucleus situated at the centre with the electron revolving around it in. He applied quantum theory in considering the energy of an electron bound to the nucleus. I would encourage you to explore further. Bohr developed a model for hydrogen atom and hydrogen like oneelectron species (hydrogenic species). 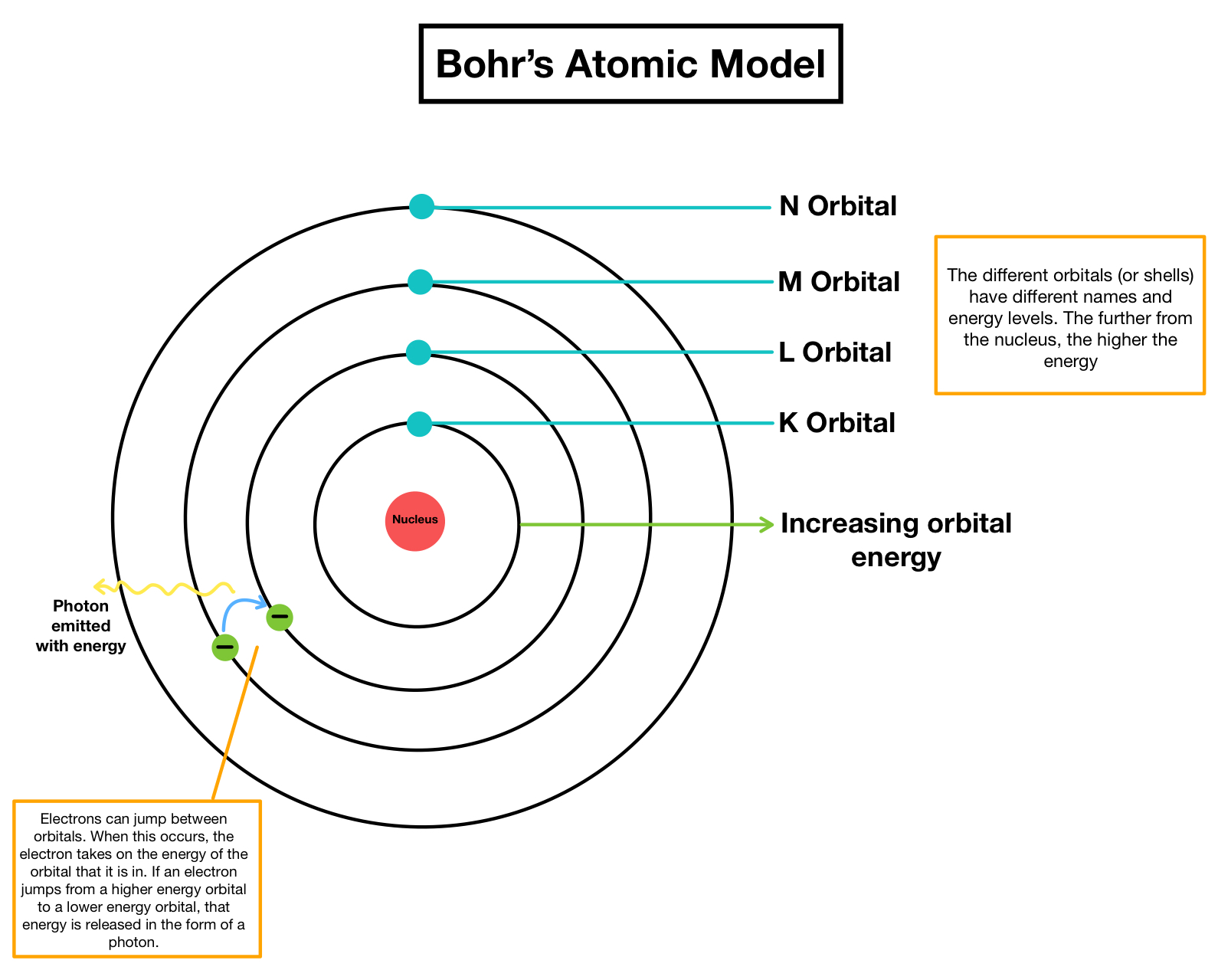
This means that radiations possess both wave-like and particle-like. One of the concepts that played a major role in the formulation of Bohr’s model is the dual nature of electromagnetic radiation. Through his experiments, the scientist Neils Bohr improved upon Rutherford’s model of the atom. looking for relationships in a meaningful way. Development Leading to Bohr’s Model of Atom. We can, however, say that the more electrons there are in an atom, the greater the variety of photon energies there will be so this may be a link worth exploringĪs I say, you are asking a great question. Bohr model of an atom consists of a small nucleus that contains protons and neutrons, this nucleus is surrounded by different electron shells or energy levels where electrons are revolved in a definite circular path similar to the structure of. as it switches from one energy level to another, then it emits / absorbs photons. Bohr model or RutherfordBohr diagram, presented by Niels Bohr and Ernest Rutherford in 1913. This is because the electron can exist in many energy levels. Atoms are way too small to see with the naked eye (and even most microscopes). 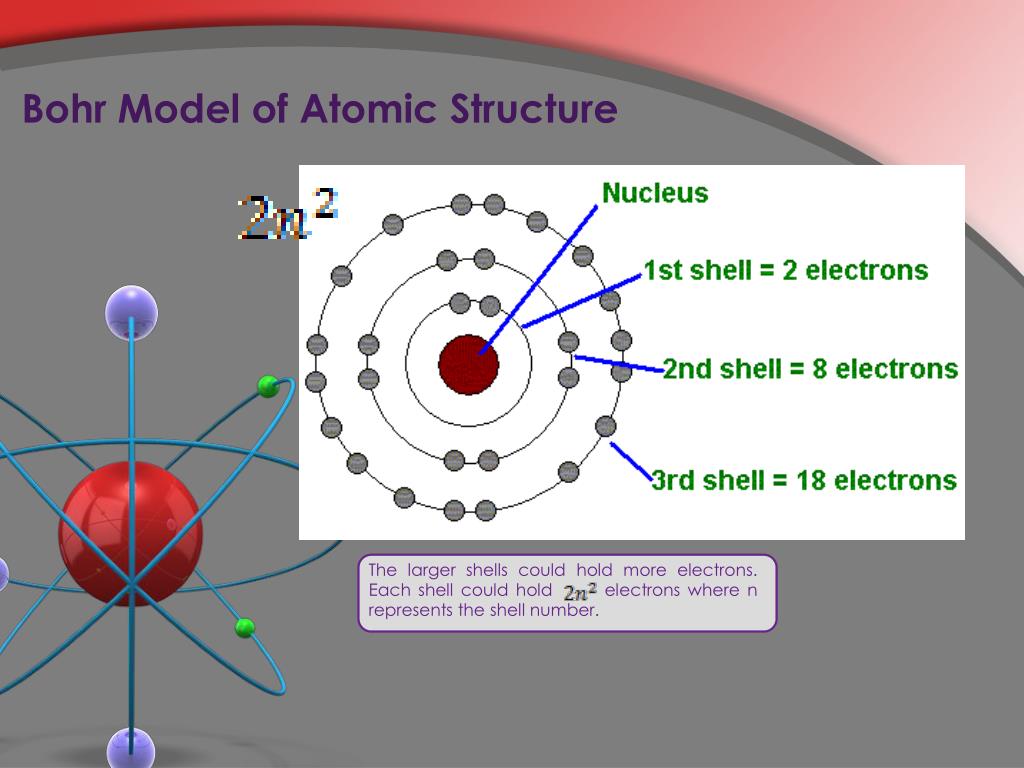
Hydrogen, for example, although the simplest atom, has a whole range of photon energies that it emits. The Bohr model Google Classroom Learn how Bohr models are used to represent atoms. The value of the energies of photons does depend on the available energy levels in the atom. Now, the energy of the photon emitted from any element does not depend on the number of electrons in the atom. OK: I would say that the periodic table tells us about the number of protons in an element and, therefore the number of electrons too. The theory of atomic structure proposed by the young Danish physicist Niels Bohr in 1913 marked the true beginning of modern atomic and quantum physics. Here is my answer, but I would encourage you to explore this and similar questions further. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
